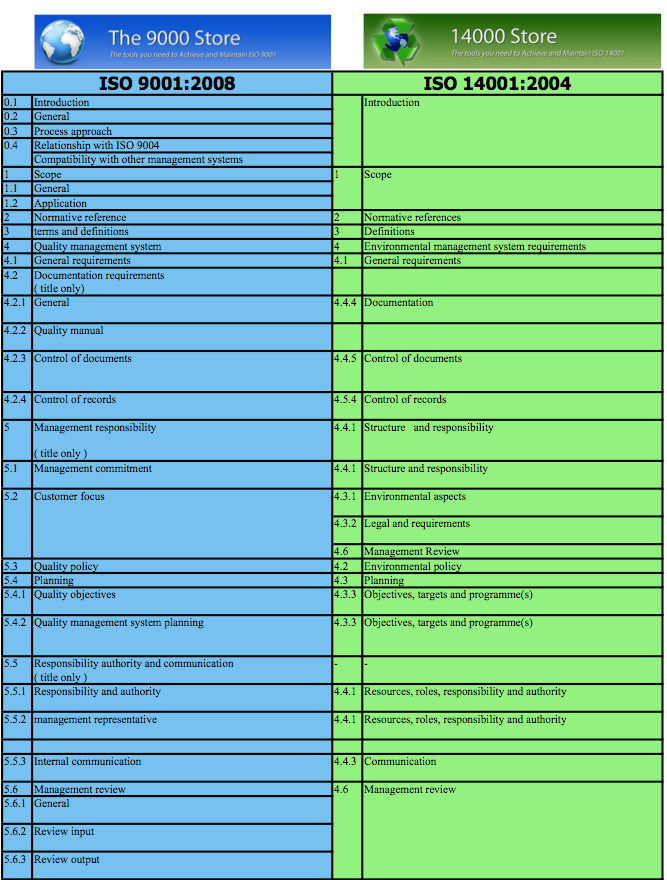
ISO 13485 is specifically targeted towards companies involved in the design, manufacturing, or supply chain processes of medical devices. 
ISO 9001 is generally the more applicable standard designed to guide any quality management system regardless of industry or business type. With the recent updates to ISO 9001: 2015 and ISO 13485: 2016, many companies are now beginning to review which standard is most applicable to their business.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
